To ROX or not to ROX? That is the question. I frequently see people confused about what ROX is and why it is used in qPCR. So, in this article, I will summarise what ROX is and why you should consider using it.
Mastering qPCR
A video tutorial to understand why to use ROX in qPCR can be found in our Mastering qPCR course.
>>Use code 20QPCR to get 20% off<<
What is ROX?
ROX, also known as carboxyrhodamine, is an inert fluorescent dye that can be added to the qPCR master mix. Unlike reporter dyes in qPCR (e.g. SYBR Green), ROX is referred to as a passive dye, since its fluorescence signal is not influenced by the PCR reactions. In other words, the fluorescent signal of ROX remains stable throughout the qPCR process and it is not related to the amount of template in the reaction.
What is the point of ROX in qPCR?
Since ROX is a passive fluorescent dye, the main reason it is used in qPCR is for signal normalisation. By normalising the fluorescent signal, this can reduce the variability between technical replicates.
Slight variations between wells can affect the fluorescent signal generated in qPCR. For example, differences in reaction volumes due to pipetting errors can bias the results. Also, issues associated with the qPCR instrument, such as the optics, can also influence the fluorescent signal generated.
You can think of the normalisation process when using ROX as a similar concept as to why reference genes are used during relative gene expression calculations, such as the Delta-Delta Ct method. Instead of controlling for the amount of template in the sample, ROX is normalising for the attributes of the qPCR mixture, such as volume differences, presence of bubble etc.
How ROX is used
To normalise the fluorescent signal, the signal of the reporter dye is divided by the signal of ROX. The new value is then known as the normalised reporter value (Rn).
Usually, in amplification plots, the delta (Δ) Rn is plotting on the y-axis against the cycle number on the x-axis. The ΔRn is the difference between the Rn of each experimental sample and the Rn of the baseline. Therefore, only the results which are above the baseline/background signal will be plotted.
 Is ROX essential for qPCR?
Is ROX essential for qPCR?
The short answer is, no. You don’t have to add ROX to your qPCR mixture. However, adding it can help reduce variations of fluorescent values in samples, which will ultimately reflect on the gene expression results.
Another important point is that the use of ROX is dependent on the qPCR you are using. Always check beforehand to see if your qPCR is compatible with ROX measurements. Some machines also prefer reactions with high ROX, while others prefer those with low ROX.
Ensure to set up the qPCR machine correctly
If you do decide to use qPCR in your reaction mixture, don’t forget to ensure the option to record ROX fluorescence as the passive dye is enabled. Otherwise, the machine will not record any data for this channel.
Similarly, if you don’t decide to use ROX, do not enable any channels which can measure ROX. Simply, the machine could think ROX is present and record background noise, which can impact on your results due to incorrect normilisation.

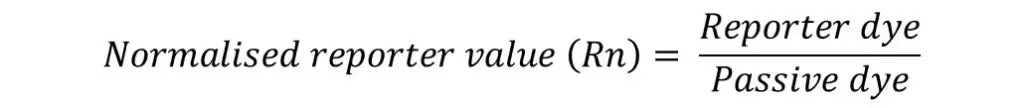
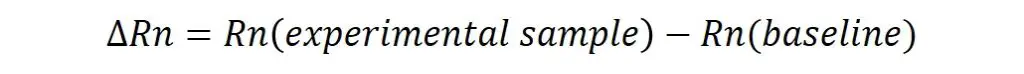



Thanks for your useful article. Zuzana
Really very important and highly informative. Thanks Dr Steven.
Really, Thanks so much… Very useful videos… So informative channel. Hope I can buying you a coffee ☕
Hi Eman,
Many thanks, I am glad you found it useful.
Thanks,
Steven